This page provides retail and manufactured food facilities with the basic label requirements which represent minimum standards for labeling food products within the State of Georgia. The Georgia Department of Agriculture (GDA) reviews product labels for formatting only.
The U.S. Food & Drug Administration (FDA) has strict labeling requirements for products that travel in interstate commerce. Any products shipped across state lines, either from the product manufacturer or as a result of secondary or tertiary distribution through a wholesale distribution point, shall fully comply with the following Federal regulations: Federal Food, Drug, and Cosmetic Act (FD&C Act), Fair Packaging and Labeling Act, Nutrition Labeling and Education Act (NLEA), and the Food Allergen Labeling and Consumer Protection Act (FALCPA).
Display Panels
Principle Display Panel (PDP)
The PDP is that portion of the package label that is most likely to be seen by the consumer at the time of purchase. Many containers are designed with two or more different surfaces that are suitable for display as the PDP. The PDP must include the product’s Statement of Identity and Declaration of Quantity.
Information Panel
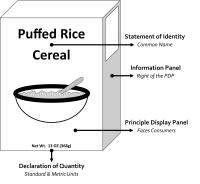
The information panel is located immediately to the right of the PDP, as displayed to the consumer. If this panel is not usable due to package design or construction (e.g., folded flaps), then the information panel is the next label panel immediately to the right. Label elements generally required to be placed together should appear on the information panel, if such labeling does not already appear on the PDP. These elements include the Declaration of Responsibility, Ingredients Statement, Nutrition Facts and Allergen Declaration.
Basic Label Elements
The following information must appear on all food labels:
1. Statement of Identity
This is the common or usual name of the product, in English, which clearly identifies what the product is.
2. Declaration of Quantity
This is the measure of the amount of food in the package or container (not including the weight of the packaging or container). It must be displayed in the lower third of the label. It must appear in both standard and metric units, and be expressed in the correct units of measure, weight (oz/g) or volume (fl oz/mL).
3. Nutrition Facts
The nutritional content must appear on the label. Nutrition facts are also required for are any health claims on the label (e.g., "Low fat," or "Low Sodium"). FDA provides an exemption of the nutrition facts requirement for small businesses.
4. Ingredients Statement
The product ingredients should be listed in order of descending predominance by weight. The ingredients statement must begin with the word "Ingredients," and all ingredients must be listed and broken down into their constituent parts. All colors, preservatives, and ingredients that are major allergens must be identified.
5. Allergen Declarations
There are 9 major food allergens that have to be clearly identified on the label. They are: MILK, EGGS, FISH, CRUSTACEAN SHELLFISH, TREE NUTS, WHEAT, PEANUTS, SESAME, and SOYBEANS. These allergens must appear in the ingredients statement, or in a “Contains:” statement immediately following the ingredients statement.
6. Handling Statement
If the product requires refrigeration for time and temperature control for safety (TCS), the label must include the phrasing: “Keep Refrigerated” or “Keep Frozen.”
7. Production Code
A product code indicating the manufacturing date must appear on the label, and/or on the package or container, in the event of a product recall.
8. Declaration of Responsibility
This is the physical address of the manufacturer. If a co-packer is used, the address must be preceded by the phrase “Manufactured for” or “Distributed by.”

More Information
UGA's Department of Food Science and Technology has information online for labeling and starting a new food business. They will also provide a nutrition facts panel for your label (for a nominal fee). See https://www.caes.uga.edu/.
The FDA has guidance documents for industry available online, which provide more detailed information and labeling requirements. See https://www.fda.gov/regulatory-information/search-fda-guidance-documents/guidance-industry-food-labeling-guide.
USDA: find information for labels as well as the WIC Program, Organics, and Country of Origin Labeling at https://www.usda.gov/.


